Isotopes And Average Atomic Mass Worksheet Answers
Calculate the actual atomic mass of 65cu. 8556 amu 2 uranium has three common isotopes.
24mg 7870 25mg 1013 and 26mg 117.
Isotopes and average atomic mass worksheet answers. The average atomic mass between these two isotopes is 63546 amu. Showing top 8 worksheets in the category isotopes and average atomic mass. 9889 12 c mass 12 amu by definition and 111 13 c mass 13003355 amu.
Hydrogen is 99 1h 08 2h and 02 3h. Carbon is composed primarily of two isotopes. Average atomic mass worksheet solutions 1 rubidium has two common isotopes 85rb and 87rb.
Calculate borons atomic mass. 75 x 133 9975 20 x 132 264 05 x 134 total 13285 amu average atomic mass determine the average atomic mass of the following mixtures of isotopes. This number is a weighted average of the masses of each of the isotopes of an element.
Isotopes and average atomic mass. The average atomic mass is the weighted average of all the isotopes of an element. 7 magnesium consists of three naturally occurring isotopes.
For example the atomic mass of carbon is reported as 12011 amu. The atomic mass is calculated from the relative abundance and the masses for these two isotopes. The percent abundance of these isotopes is as follows.
Isotopes and average atomic mass as concepts allow for the specific discussion of elements and their atoms and this quizworksheet combo will help you test your understanding of these concepts. Some of the worksheets displayed are chemistry average atomic mass work average atomic mass problems key 2013 average atomic mass practice problems abundance of isotopes name chem work 4 3 atomic particles atoms isotopes and bonding work isotopic abundance practice problems. A sample of cesium is 75 133cs 20 132cs and 5 134cs.
Because most elements exist as mixtures of several stable isotopes the atomic mass of an element is defined as the weighted average of the masses of the isotopes. Calculate its average atomic mass. What is its average atomic mass.
Rubidium is a soft silvery white metal that has two common isotopes 85rb and 87rb. For example naturally occurring carbon is largely a mixture of two isotopes. If the abundance of 85rb is 722 and the abundance of 87rb is 278 what is the average atomic mass of rubidium.
If the abundance of 85rb is 802 and the abundance of 87rb is 198 what is the average atomic mass of rubidium.
 Rontavstudio Phet Isotopes And Atomic Mass Worksheet Answers
Rontavstudio Phet Isotopes And Atomic Mass Worksheet Answers
 29 Inspirational Isotopes And Average Atomic Mass Worksheet Images
29 Inspirational Isotopes And Average Atomic Mass Worksheet Images
Calculating Atomic Mass Worksheet Small Size Chemistry Calculating

 Isotope Practice Worksheet Answer Key Best Of Average Atomic Mass
Isotope Practice Worksheet Answer Key Best Of Average Atomic Mass
 Average Atomic Mass Worksheet By Ms Stricklin Chemistry Corner Tpt
Average Atomic Mass Worksheet By Ms Stricklin Chemistry Corner Tpt
 Average Atomic Mass Copper Is Made Of Two Isotopes Copper 63
Average Atomic Mass Copper Is Made Of Two Isotopes Copper 63
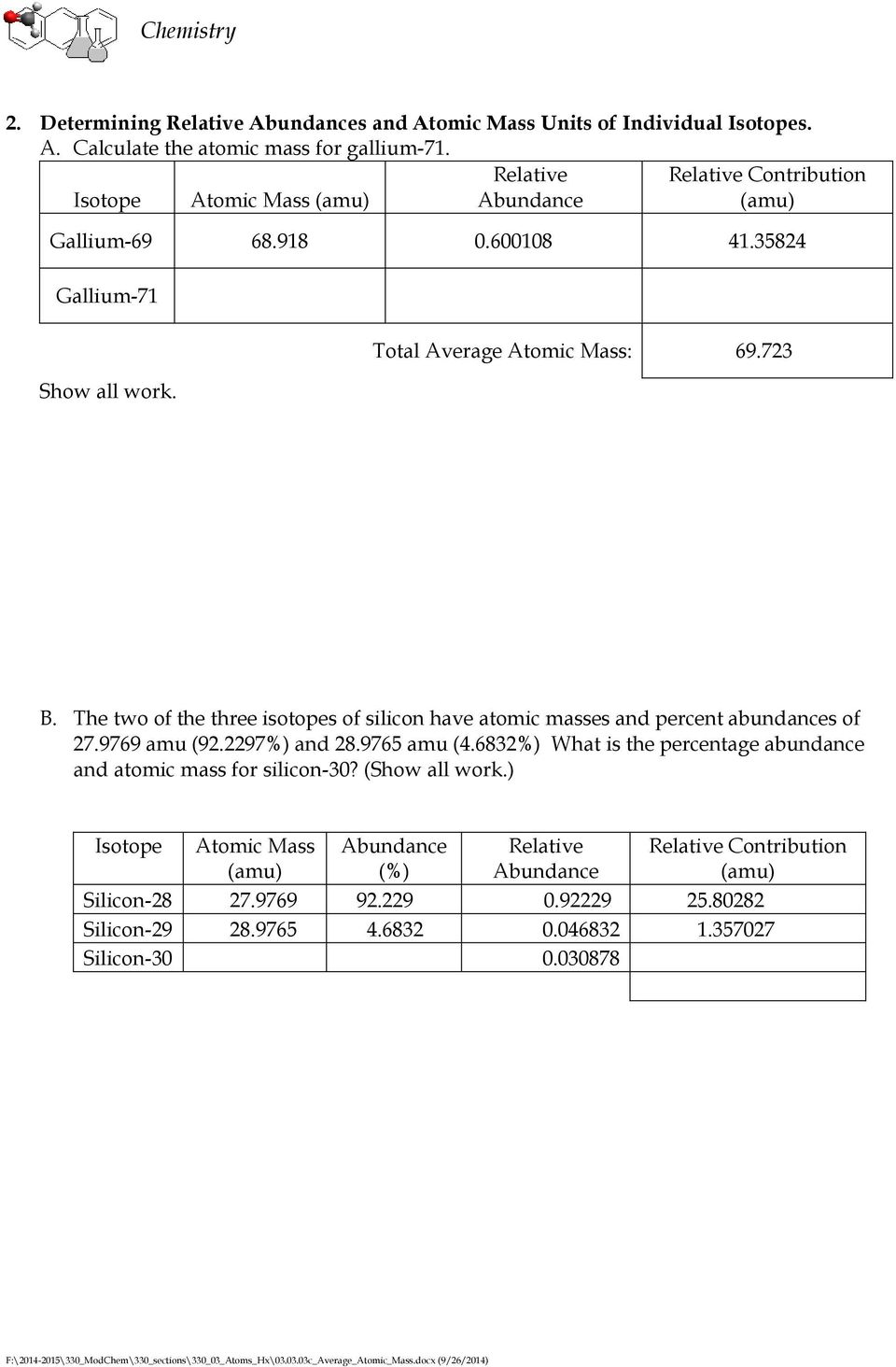 Determining Average Atomic Mass Pdf
Determining Average Atomic Mass Pdf
 Isotopes And Average Atomic Mass Worksheet Answers Lobo Black
Isotopes And Average Atomic Mass Worksheet Answers Lobo Black
 Answer An Element Has Three Isotopes Giv Clutch Prep
Answer An Element Has Three Isotopes Giv Clutch Prep
 Average Atomic Mass And Isotopes The Atomic Mass Of Vegium Lab
Average Atomic Mass And Isotopes The Atomic Mass Of Vegium Lab
 Quiz Worksheet Isotopes And Average Atomic Mass Study Com
Quiz Worksheet Isotopes And Average Atomic Mass Study Com
Calculating Atomic Mass Worksheet Isotopes And Average Atomic Mass
 Isotopes And Average Atomic Mass Worksheet Answers Briefencounters
Isotopes And Average Atomic Mass Worksheet Answers Briefencounters
 Quiz Worksheet Average Atomic Mass Study Com
Quiz Worksheet Average Atomic Mass Study Com
Abundance Of Isotopes Chem Worksheet 4 3 Super New Average Atomic
 Periodic Table With Atomic Masses Inspirational Average Atomic Mass
Periodic Table With Atomic Masses Inspirational Average Atomic Mass
 Model 2 Mix Isotopes Play With The Mix Isotopes Tab For A Few
Model 2 Mix Isotopes Play With The Mix Isotopes Tab For A Few
 Isotopes And Average Atomic Mass Worksheet Answer Key Archives
Isotopes And Average Atomic Mass Worksheet Answer Key Archives
 Solved Worksheet Isotope Problems 1 A Student Looked Up
Solved Worksheet Isotope Problems 1 A Student Looked Up
 Determination Of Average Atomic Mass Worksheet For 10th Grade
Determination Of Average Atomic Mass Worksheet For 10th Grade
Isotopes And Average Atomic Mass Worksheets Answers
Average Atomic Mass Worksheet Periodic Table Protons Copy Neutrons
Average Atomic Mass Worksheet Holidayfu Com
 Isotopes And Average Atomic Mass Worksheet Awesome Free Worksheets
Isotopes And Average Atomic Mass Worksheet Awesome Free Worksheets
All Worksheets Average Atomic Mass Worksheet Answers Isotopes And





0 Response to "Isotopes And Average Atomic Mass Worksheet Answers"
Post a Comment